Nipah Virus: A Scientific and Epidemiological Deep Dive into a Persistent Pandemic Threat
Abstract
Nipah virus (NiV) is a zoonotic paramyxovirus of the genus Henipavirus that has emerged as a pathogen of critical global health concern. Characterized by severe encephalitic and respiratory manifestations, high case-fatality ratios (CFRs), and documented human-to-human transmission, NiV exemplifies the formidable challenges posed by bat-borne emerging infectious diseases (EIDs). Since its initial identification in 1999, recurrent outbreaks, primarily in South and Southeast Asia, have underscored its epidemic potential and the urgent need for robust countermeasures. This article provides a comprehensive scientific overview of NiV, encompassing its virology, ecology, epidemiology, clinical pathogenesis, diagnostic modalities, current management strategies, and the multifaceted global efforts in prevention, preparedness, and research.
1. Introduction: The Age of Emergence
The 21st century has been defined by the recurrent emergence and re-emergence of infectious diseases with pandemic potential. Factors such as ecological disruption, climate change, intensification of agriculture, and globalized travel have created a nexus for viral spillover from animal reservoirs into human populations. Among these threats, the Nipah virus stands out not only for its alarming virulence but also as a paradigm of the complex interplay between wildlife, livestock, and human health. Classified as a WHO R&D Blueprint Priority Pathogen and a CDC Category C Bioterrorism Agent, NiV represents a clear and present danger to global health security, necessitating a sophisticated understanding of its biology and ecology to mitigate future outbreaks.
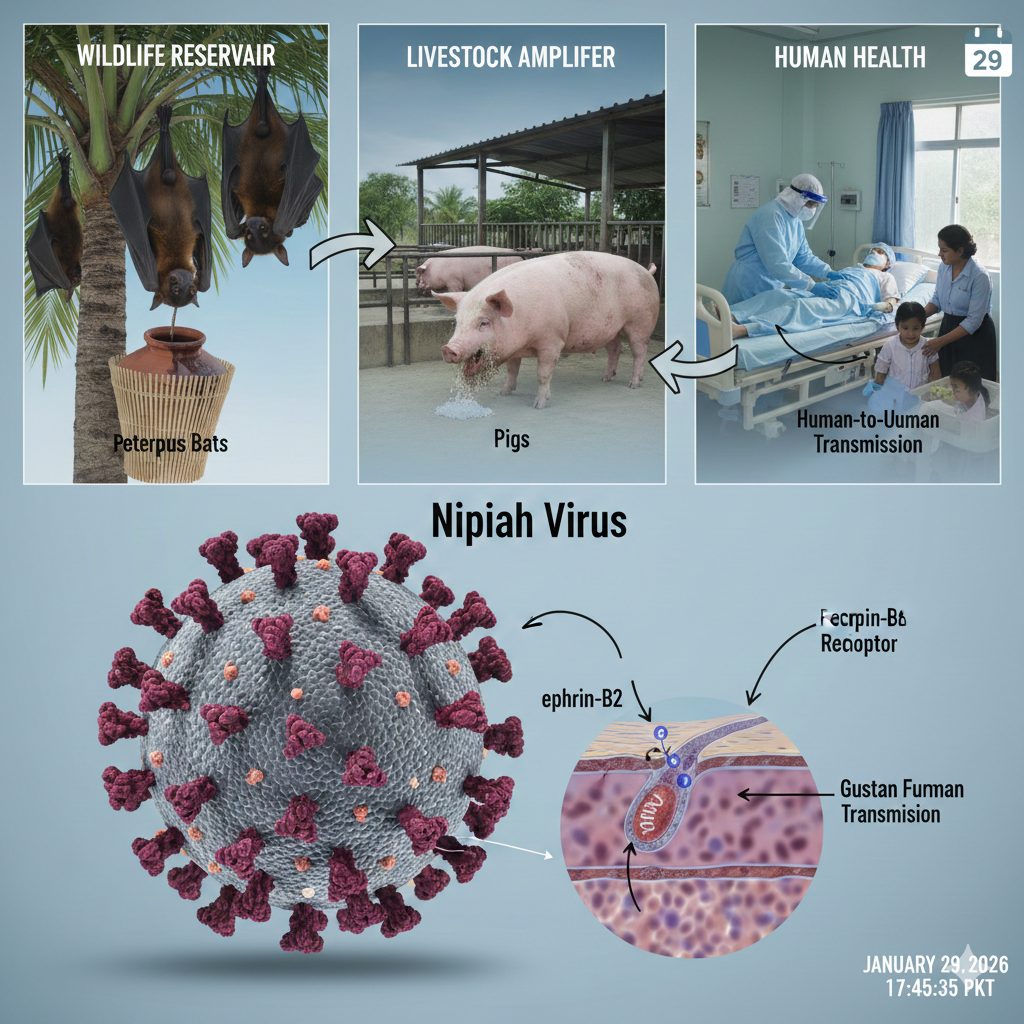
2. Virology: The Molecular Architecture of a Pathogen
Taxonomy and Structure:
Nipah virus is an enveloped, negative-sense, single-stranded RNA virus belonging to the family Paramyxoviridae, genus Henipavirus. Its closest relative is the Hendra virus (HeV), with which it shares significant genetic and structural homology.
Genome:
Approximately 18.2 kilobases in length, the NiV genome encodes six major structural proteins: nucleocapsid (N), phosphoprotein (P), matrix (M), fusion (F), glycoprotein (G), and large polymerase (L). The P gene undergoes complex transcriptional editing to produce accessory proteins (V, W, C) that are critical virulence factors, primarily through their role as potent antagonists of the host innate immune response, particularly the interferon (IFN) signaling pathway.
Surface Glycoproteins:
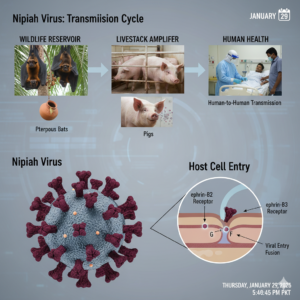
The F (fusion) and G (attachment) glycoproteins form the functional unit for host cell entry. The G protein binds to host cell receptors, while the F protein mediates membrane fusion. A key determinant of NiV’s broad species tropism and pathogenicity is the use of highly conserved cell surface ephrin-B2 and ephrin-B3 proteins as primary entry receptors. These receptors are ubiquitously expressed in endothelial cells, neurons, and smooth muscle, explaining the virus’s proclivity for causing widespread vasculitis and encephalitis.
Viral Diversity:
Two major NiV strains have been characterized:
- NiV-Malaysia (NiV-M): The prototype strain from the 1998-99 outbreak. It displayed a strong porcine–human transmission route and lower observed human-to-human transmissibility.
- NiV-Bangladesh (NiV-B): The strain responsible for recurrent outbreaks in Bangladesh and India. It demonstrates more efficient human-to-human transmission and may be associated with distinct clinical presentations, including acute respiratory distress syndrome.
3. History and Epidemiology: Tracing the Spillover Events
The Index Outbreak (1998-1999, Malaysia & Singapore):
The virus emerged in the pig-farming communities of Negeri Sembilan, Malaysia. A simultaneous outbreak of severe encephalitis among pigs and humans was initially misattributed to Japanese encephalitis. However, the epidemiological pattern—affecting adult pig farmers rather than children—and the failure of mosquito control and JE vaccination prompted further investigation. In March 1999, scientists at the University of Malaya isolated a novel virus, later named after Sungai Nipah village. The outbreak resulted in 265 human cases and 105 deaths (CFR ~40%). The economic impact was devastating, with the culling of over one million pigs to contain the virus. This event highlighted the risks of agricultural intensification at the forest-livestock interface.
The Evolving Pattern in South Asia:
Subsequent outbreaks revealed a different transmission dynamic, centered on Bangladesh (annually since 2001) and India (West Bengal in 2001, 2007; Kerala in 2018, 2019, 2021, 2023).
- Bangladesh: Outbreaks are seasonal (December-May), tightly linked to the consumption of raw date palm sap (tari or khejurer rosh). Sap collection pots are contaminated overnight by fruit bat urine or saliva. This represents a classic foodborne zoonotic route. These outbreaks have shown higher CFRs (often exceeding 70%) and more frequent human-to-human transmission, particularly in hospital and household settings.
- India – The Kerala Model: The 2018 outbreak in Kozhikode, Kerala, was a stark reminder of NiV’s epidemic potential in a region with a high population density and excellent healthcare connectivity. Despite a high initial CFR, the outbreak was contained through an exceptionally rapid and coordinated public health response: aggressive contact tracing, strict isolation protocols, establishment of containment zones, and effective risk communication. Kerala’s success has been lauded as a model for outbreak response.
Geographical Range and At-Risk Regions:
The natural reservoir, Pteropus bats, are distributed from the western Indian Ocean through South and Southeast Asia to the Pacific Islands. Therefore, over two billion people live in regions where the reservoir hosts and risk factors (e.g., palm sap consumption, livestock farming) overlap, making NiV a pervasive, if sporadic, threat.
4. Ecology and Reservoir Hosts: The Silent Carriers
Flying Foxes (Genus Pteropus):
These large fruit bats (megabats) are the confirmed natural reservoir for NiV. They exhibit a persistent, asymptomatic infection, with viral shedding in urine, saliva, and feces. The virus maintains itself in bat populations through a metapopulation dynamic, with periodic epizootics and seasonal shedding patterns.
Spillover Mechanisms:
The disruption of bat habitats due to deforestation, urbanization, and agricultural expansion is hypothesized to increase stress-induced viral shedding in bats and force them into closer proximity with human settlements and farms. Spillover occurs via:
- Direct Contamination: Bat-contaminated fruits or date palm sap.
- Amplifying Hosts: As seen in Malaysia, pigs act as efficient amplifying hosts. Upon infection, pigs develop a severe respiratory illness, shedding massive amounts of virus in respiratory secretions and bodily fluids, leading to rapid farm-wide epizootics and high-risk exposure for farmers.
5. Transmission Dynamics: From Bats to Humans and Beyond
Primary Zoonotic Transmission:
- Consumption of Contaminated Foods: The primary route in Bangladesh.
- Direct Contact with Bats: Handling sick bats or their products.
- Exposure via Infected Livestock: Direct contact with tissues, secretions, or aerosols from sick pigs, horses, or other domestic animals.
Secondary Human-to-Human Transmission:
This is the most significant factor for epidemic potential. Transmission occurs via direct contact with infectious bodily fluids (respiratory droplets, saliva, urine, blood). Documented high-risk scenarios include:
- Caregiving: Caring for infected family members without adequate personal protective equipment (PPE).
- Hospital-Acquired (Nosocomial) Transmission: A critical amplifier in past outbreaks. Infected patients presenting with respiratory symptoms can infect healthcare workers and other patients, leading to devastating hospital-based clusters.
Incubation Period:
Typically 4-14 days, but reports extend up to 45 days. This prolonged, variable incubation period poses immense challenges for surveillance, contact tracing, and quarantine efficacy.
6. Clinical Manifestations and Pathogenesis
NiV infection can range from asymptomatic (documented in a significant proportion of cases during serosurveys) to acute lethal encephalitis.
Clinical Spectrum:
Asymptomatic Infection
Estimated to occur in a subset of exposed individuals; its role in transmission is not fully understood.
Acute Febrile Encephalitic Syndrome
The classic presentation. After a non-specific prodrome of fever, headache, myalgia, and vomiting, rapid neurological deterioration ensues within 24-48 hours.
Atypical Presentations
NiV-B strain outbreaks have shown a higher incidence of severe acute respiratory distress syndrome (ARDS), often preceding neurological signs.
Relapsing/Late-Onset Encephalitis
A unique and devastating feature reported in 3-10% of survivors. Neurological symptoms can recur months or even years after the initial infection.
Pathogenesis:
The virus enters via the respiratory or gastrointestinal tract, replicates locally, and disseminates hematogenously (viremia). The binding of the G protein to ephrin-B2/B3 receptors on vascular endothelial cells leads to widespread vasculitis, endothelial syncytia formation, and increased vascular permeability. This results in ischemia, hemorrhage, and subsequent parenchymal damage in organs rich in these receptors, notably the brain (encephalitis) and lungs (pneumonia). The immune evasion strategies mediated by the V, W, and C proteins contribute to uncontrolled viral replication and severe disease.
Case Fatality and Sequelae:
CFR varies by outbreak and strain (40%-75%, up to 100% in some small clusters). Survivors of acute encephalitis face a high burden of long-term neurological sequelae, including persistent convulsions, personality changes, residual paresis, and cognitive impairment.
7. Diagnosis: High-Consequence Laboratory Testing
Clinical diagnosis is difficult due to non-specific early symptoms. Laboratory confirmation is mandatory and conducted under high-containment biosafety conditions (BSL-4 for virus culture, BSL-3 for molecular assays).
Key Diagnostic Modalities:
| Method | Type | Description |
|---|---|---|
| Real-Time RT-PCR | Molecular | The gold standard for early detection. Can be performed on throat/nasal swabs, blood, CSF, or urine. |
| ELISA | Serological | Detects IgM (acute infection) and IgG (past infection) antibodies. Often used for serosurveillance. |
| Virus Neutralization Test | Serological | The definitive serological test, measuring neutralizing antibodies. Requires BSL-4 facilities. |
| Viral Isolation | Culture | Attempted only in reference BSL-4 laboratories from clinical samples. |
8. Management and Treatment: The Critical Care Paradigm
⚠️ No licensed specific antiviral therapy or vaccine exists for human use. Management is entirely supportive and intensive.
Supportive Care
- Neurological Support: Aggressive management of raised intracranial pressure, anti-epileptics for seizures
- Respiratory Support: Early recognition and management of hypoxia, often requiring mechanical ventilation for ARDS
- Systemic Support: Management of sepsis, fluid/electrolyte balance, and prevention of secondary infections
Investigational Therapeutics
- Monoclonal Antibodies (mAbs): m102.4 is the most promising candidate, targeting the ephrin-binding site of NiV G glycoprotein.
- Broad-Spectrum Antivirals: Ribavirin (anecdotal benefit) and Remdesivir (in vitro and animal model efficacy).
- Fusion/Entry Inhibitors: Peptide fusion inhibitors and small molecules targeting F and G proteins in preclinical development.
9. Prevention, Control, and Public Health Strategy
Given the absence of specific medical countermeasures, prevention is the cornerstone of NiV management.
1. Primary Prevention (Reducing Spillover Risk):
- Date Palm Sap Safety: Public education to avoid raw sap consumption. Physical barriers to prevent bat access.
- Agricultural and Livestock Biosecurity: Preventing bat access to farms, using protective gear, establishing buffer zones.
- Food Safety: Washing and peeling fruits, cooking potentially contaminated foods.
2. Interrupting Human-to-Human Transmission:
- Infection Prevention and Control (IPC): Critical in healthcare settings. Includes strict precautions, patient isolation, PPE use.
- Community Engagement: Educating communities on early symptoms, dangers of unprotected caregiving, safe burial practices.
3. Surveillance and Outbreak Response:
- Integrated One Health Surveillance: Coordinating human, animal, and environmental surveillance.
- Rapid Response Teams: Pre-identified, trained multidisciplinary teams for case investigation and contact tracing.
- Risk Communication: Transparent, timely, and culturally appropriate communication.
10. Global Preparedness and Research Frontiers
NiV is a prototype pathogen for pandemic preparedness due to its RNA genome, capacity for mutation, and proven human-to-human transmission.
WHO Blueprint and CEPI:
The Coalition for Epidemic Preparedness Innovations (CEPI) has prioritized NiV vaccine development. Several platforms are in preclinical and early clinical stages:
- Viral Vector Vaccines: Using platforms like ChAdOx1, MVA, or VSV expressing NiV F or G glycoproteins.
- Subunit Vaccines: Recombinant soluble G or F proteins.
- mRNA Vaccines: Leveraging the platform validated during the COVID-19 pandemic.
Research Priorities:
• Ecological drivers of viral shedding
• Point-of-care diagnostic tests
• Clinical trials for therapeutics
• Mechanisms of viral persistence
11. Conclusion: A Persistent Threat Requiring Sustained Vigilance
The Nipah virus is not merely a regional health issue but a global biosafety and security concern. It embodies the complex challenges of the Anthropocene: ecological disruption, food security, and pandemic vulnerability. While scientific progress in therapeutics and vaccines is encouraging, the current defense rests on a foundation of robust public health infrastructure, effective One Health collaboration, and community-led prevention. The lessons from Kerala and ongoing research offer a blueprint for containment and hope. Ultimately, mitigating the threat of Nipah virus requires a long-term, integrative commitment to understanding the delicate balance between human, animal, and environmental health, ensuring we are better prepared for the next inevitable spillover event.
Preparedness today prevents pandemics tomorrow